Federal judge allows continued mifepristone distribution while ordering FDA safety review
A federal judge ruled the abortion pill mifepristone can remain available by mail nationwide while requiring the FDA to conduct a safety review.
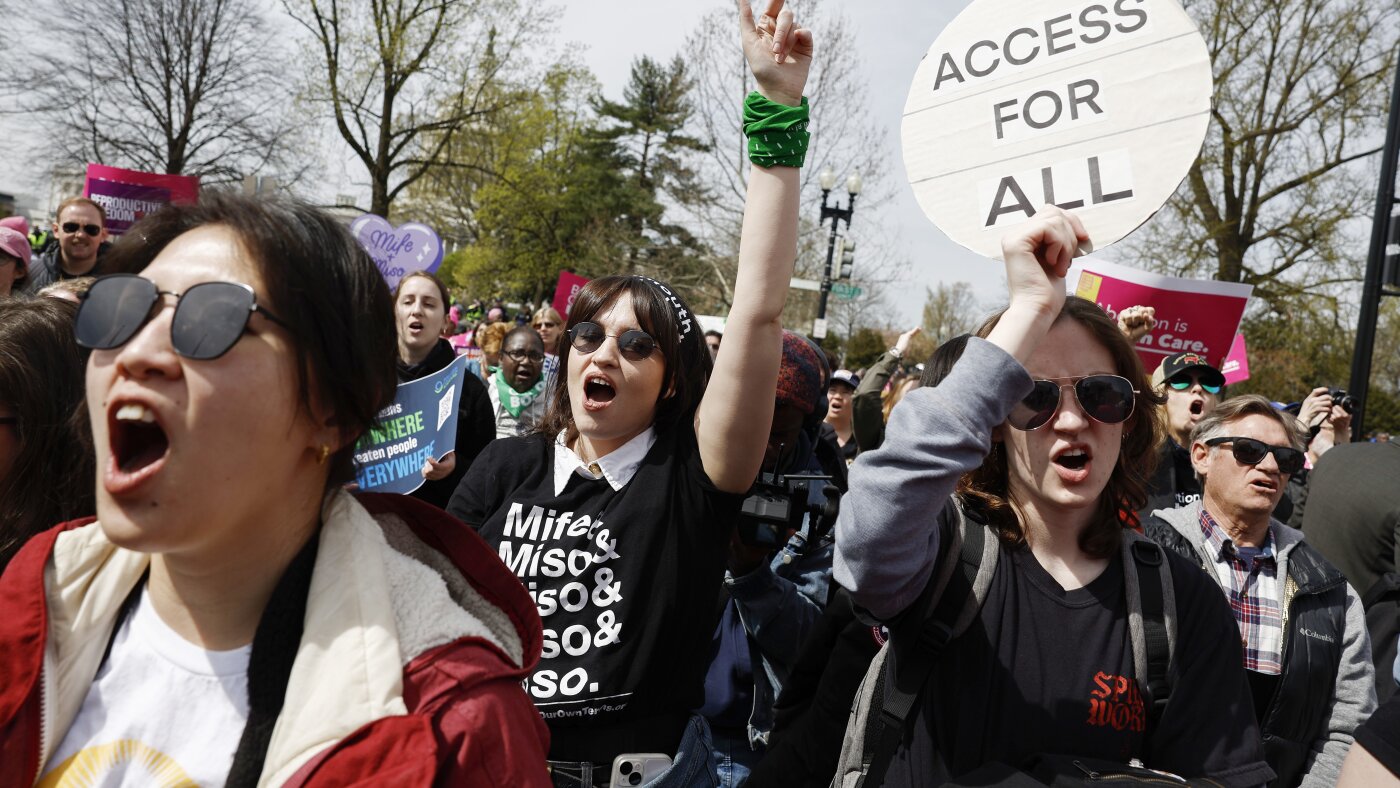
A federal judge has ruled that the abortion pill mifepristone can continue to be distributed nationwide by mail while ordering the Food and Drug Administration to conduct a safety review of the medication.
The ruling addresses a case brought by Louisiana seeking to ban the use of mifepristone through telemedicine services. The judge determined that while the medication can remain available through current distribution channels for now, the FDA must complete a comprehensive safety review before the case can proceed further.
The decision means that patients will continue to have access to mifepristone through existing telehealth and mail-order systems in the immediate term. The medication is commonly used in combination with another drug, misoprostol, for medication abortions and is approved by the FDA for use up to 10 weeks of pregnancy.
The judge indicated that significant changes to the FDA's current guidelines for mifepristone could be forthcoming depending on the results of the safety review. The FDA faces a six-month deadline to complete this review process.
The Louisiana case represents one of several ongoing legal challenges to abortion access following changes in federal and state abortion laws. The outcome of the FDA review and subsequent court proceedings could affect how the medication is prescribed and distributed across the United States.